Periodicity Reference Sheet
ADVERTISEMENT
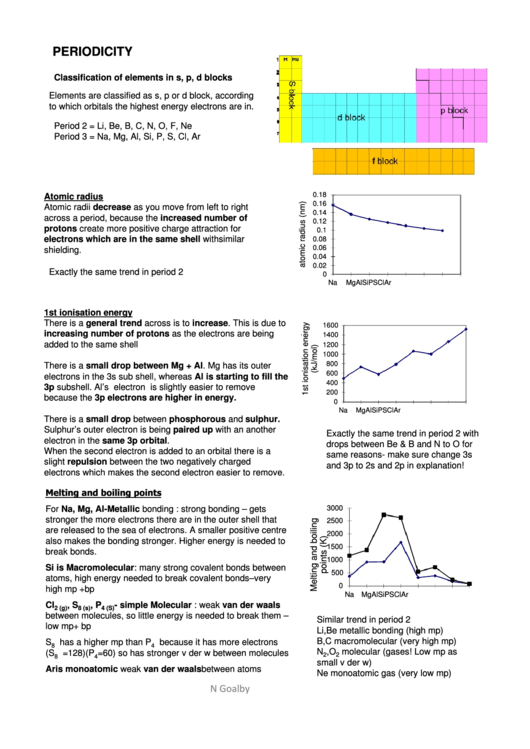
PERIODICITY
Classification of elements in s, p, d blocks
Elements are classified as s, p or d block, according
to which orbitals the highest energy electrons are in.
Period 2 = Li, Be, B, C, N, O, F, Ne
Period 3 = Na, Mg, Al, Si, P, S, Cl, Ar
0.18
Atomic radius
0.16
Atomic radii decrease as you move from left to right
0.14
across a period, because the increased number of
0.12
protons create more positive charge attraction for
0.1
electrons which are in the same shell with similar
0.08
0.06
shielding.
0.04
0.02
Exactly the same trend in period 2
0
Na
Mg
Al
Si
P
S
Cl
Ar
1st ionisation energy
There is a general trend across is to increase. This is due to
1600
increasing number of protons as the electrons are being
1400
added to the same shell
1200
1000
800
There is a small drop between Mg + Al. Mg has its outer
600
electrons in the 3s sub shell, whereas Al is starting to fill the
400
3p subshell. Al’s electron is slightly easier to remove
200
because the 3p electrons are higher in energy.
0
Na
Mg
Al
Si
P
S
Cl
Ar
There is a small drop between phosphorous and sulphur.
Sulphur’s outer electron is being paired up with an another
Exactly the same trend in period 2 with
electron in the same 3p orbital.
drops between Be & B and N to O for
When the second electron is added to an orbital there is a
same reasons- make sure change 3s
slight repulsion between the two negatively charged
and 3p to 2s and 2p in explanation!
electrons which makes the second electron easier to remove.
Melting and boiling points
3000
For Na, Mg, Al- Metallic bonding : strong bonding – gets
stronger the more electrons there are in the outer shell that
2500
are released to the sea of electrons. A smaller positive centre
2000
also makes the bonding stronger. Higher energy is needed to
1500
break bonds.
1000
Si is Macromolecular: many strong covalent bonds between
500
atoms, high energy needed to break covalent bonds– very
0
high mp +bp
Na Mg
Al
Si
P
S
Cl
Ar
Cl
, S
, P
- simple Molecular : weak van der waals
2 (g)
8 (s)
4 (S)
between molecules, so little energy is needed to break them –
Similar trend in period 2
low mp+ bp
Li,Be metallic bonding (high mp)
B,C macromolecular (very high mp)
S
has a higher mp than P
because it has more electrons
8
4
N
,O
molecular (gases! Low mp as
(S
=128)(P
=60) so has stronger v der w between molecules
2
2
8
4
small v der w)
Ar is monoatomic weak van der waals between atoms
Ne monoatomic gas (very low mp)
N Goalby
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1