Mole Conversions Worksheet
ADVERTISEMENT
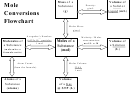
Mole
C onversions
W orksheet
There
a re
t hree
m ole
e qualities.
T hey
a re:
23
1
m ol
=
6 .02
x
1 0
p articles
1
m ol
=
g -‐formula-‐mass
( periodic
t able)
1
m ol
=
2 2.4
L
f or
a
g as
a t
S TP
Mole-‐Particle
C onversions
22
1. How
m any
m oles
o f
m agnesium
i n
3 .01
x
1 0
atoms
o f
m agnesium?
2. How
m any
m olecules
a re
t here
i n
4 .00
m oles
o f
g lucose,
C
H
O
?
6
12
6
Mole-‐Mass
C onversions
3. How
m any
m oles
i n
2 8
g rams
o f
C O
?
2
4. What
i s
t he
m ass
o f
5
m oles
o f
F e
O
?
2
3
Mole-‐Volume
C onversions
5. Determine
t he
v olume,
i n
l iters,
o ccupied
b y
0 .030
m oles
o f
a
g as
a t
S TP.
6. How
m any
m oles
o f
a rgon
a toms
a re
p resent
i n
1 1.2
L
o f
a rgon
g as
a t
S TP?
Mixed
M ole
C onversions
7. How
m any
o xygen
m olecules
a re
i n
3 .36
L
o f
o xygen
g as
a t
S TP?
23
8. Find
t he
m ass
i n
g rams
o f
2 .00
x
1 0
m olecules
o f
F
.
2
9. Determine
t he
v olume
i n
l iters
o ccupied
b y
1 4.0
g
o f
n itrogen
g as
a t
S TP.
23
10. Find
t he
m ass,
i n
g rams,
o f
1 .00
x
1 0
m olecules
o f
N
.
2
11. Aspartame
i s
a n
a rtificial
s weetener
t hat
i s
1 60
t imes
s weeter
t han
s ucrose
( table
s ugar)
w hen
dissolved
i n
w ater.
I t
i s
m arketed
a s
N utraSweet.
T he
m olecular
f ormula
o f
a spartame
i s
C
H
N
O
.
14
18
2
5
i) Calculate
t he
g ram-‐formula-‐mass
o f
a spartame.
ii) How
m any
m oles
o f
m olecules
a re
i n
1 0
g
o f
a spartame?
iii) What
i s
t he
m ass
i n
g rams
o f
1 .56
m oles
o f
a spartame?
iv) How
m any
m olecules
a re
i n
5 .0000
m g
o f
a spartame?
v) How
m any
a toms
o f
n itrogen
a re
i n
1 .2000
g rams
o f
a spartame?
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1